How to Prevent Corrosion
Corrosion is one of the greatest adversaries that metals face. It is a natural occurrence caused by the reaction of metal and environmental factors.
Corrosion can compromise the appearance of the metal and, even worse, affect performance in terms of strength and load-bearing ability. Luckily, there are effective corrosion prevention methods that can extend the life of metal equipment by up to 250%.
Not all corrosion prevention methods are equal. It's crucial to choose the right method based on the type of metal and the environmental factors in question.
Corrosion-resistant metals
One of the most effective ways of corrosion prevention is using metals that are not prone to corrosion. These include aluminum and stainless steel.
If the application in mind allows, purchasing metals that don't corrode easily can reduce the need for additional corrosion protection, especially if the applications are not close to water bodies.
Protective Coating
Protective Coating

A fresh coat of paint will enhance the appearance of your metal structure and prevent corrosion. However, not all paints are as effective in preventing corrosion. Rubber paints are the most effective because they act as a barrier that prevents the metal's chemical structure from interacting with environmental compounds like water and oxygen that result in corrosion.
Applying a powder coat is another effective method of corrosion prevention. The powder coat applied on the surface of the metal is heated to form a smooth protective film. The most commonly used powders are nylon, urethane, polyester, epoxy, and acrylic. For metal sheets, polycoating can also help to prevent corrosion to some extent and also hide imperfection caused by contact with the rollers at the mills.
Metal Plating
Metal Plating

Theoretically, plating is almost similar to painting. Instead of paint, a thin layer of metal is applied to the metal you want to protect. The metal layer prevents corrosion and adds an aesthetic finish.
There are four types of metallic plating:
Electroplating – A thin layer of metal like chromium or nickel is deposited in the substrate metal in an electrolyte bath.
Mechanical plating – It involves cold welding a metal powder to the substrate material.
Electroless – This method uses a chemical reaction to deposit a coating of metal like nickel or cobalt to the primary metal. The method doesn't require an electric current to complete.
Hot dipping – It's a simple coating technique that requires the substrate to be immersed in a molten bath of protective metal, forming a thin protective layer around the substrate metal.
Corrosion inhibitors
Corrosion inhibitors
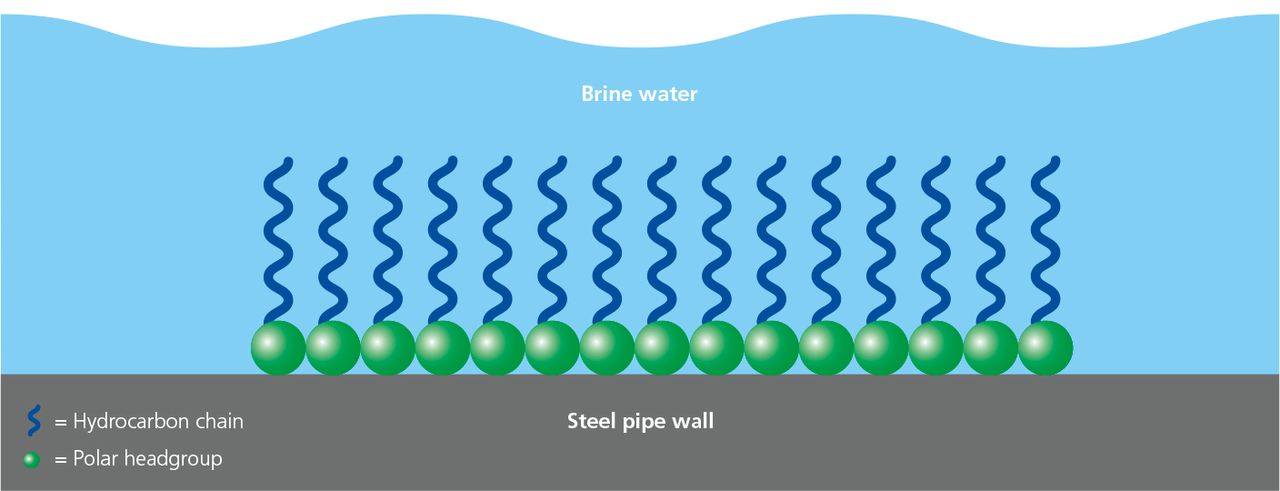
Corrosion inhibitors are chemicals applied to the surface of the metal that react with the metal or the surrounding gases to inhibit or suppress the electrochemical processes that lead to corrosion.
Sacrificial coatings
Sacrificial coatings

In this method, a coat of a metal that is likely to oxidize is added on the surface of the metal you want to protect. You can either use cathodic protection in a process known as galvanizing or use anodic protection.
Environmental Measures
Environmental Measures
The environment plays a significant role in the corrosion process. By controlling the environment, you can prevent or reduce the rate of corrosion. There are both simple and complex methods you can try.
The simple ones include reducing exposure to moisture while complex alternatives include controlling the oxygen, sulfur, or chlorine levels in the environment around the metal.
Modifying the design
Modifying the design

Designing with corrosion in mind can help inhibit the process and prolong the life of the metal. Simple adjustments like avoiding cracks and pits where the metal can hold water or encourage free movement of air can go a long way in preventing corrosion. It’s also vital to consider regular maintenance as an additional method.
One of the best ways of preventing corrosion is planning ahead. Having effective corrosion prevention methods can prolong the life of metal and retain the aesthetic appearance.


























